|
WEATHERING OF OIL AND
GAS IN THE COASTAL MARINE ENVIRONMENT: QUANTIFYING RATES OF
MICROBIAL METABOLISM
Principal Investigator:
David Valentine (UCSB)
Summary of Research
 Large quantities of oil and gas are released into
the Santa Barbara Channel by way of natural seepage with lesser amounts
emitted during petroleum removal and recovery. These emissions
greatly affect beach, air and water quality along the Southern
California Coast. As a result, many studies and a substantial
amount of resources have been devoted to developing a better
understanding of the weathering processes occurring in hydrocarbon-rich
environments. These studies have provided evidence for natural
hydrocarbon-consuming communities thriving in heavily contaminated
regions. It is assumed that native assemblages of microorganisms having
the capability to consume a variety of hydrocarbons emitted from
natural oil seeps are present in the Santa Barbara channel.
Although microbial oxidation is known to occur, little is known about
the distribution of relevant microbial communities, rates of oxidation
and the extent to which various hydrocarbons are broken down or
consumed. Large quantities of oil and gas are released into
the Santa Barbara Channel by way of natural seepage with lesser amounts
emitted during petroleum removal and recovery. These emissions
greatly affect beach, air and water quality along the Southern
California Coast. As a result, many studies and a substantial
amount of resources have been devoted to developing a better
understanding of the weathering processes occurring in hydrocarbon-rich
environments. These studies have provided evidence for natural
hydrocarbon-consuming communities thriving in heavily contaminated
regions. It is assumed that native assemblages of microorganisms having
the capability to consume a variety of hydrocarbons emitted from
natural oil seeps are present in the Santa Barbara channel.
Although microbial oxidation is known to occur, little is known about
the distribution of relevant microbial communities, rates of oxidation
and the extent to which various hydrocarbons are broken down or
consumed.
This MMS-UC CMI funded research focuses on the microbial
weathering of aromatic compounds released into marine
environments. The objectives of this research include: (1)
determing the intermediates and end products arising from microbial
decomposition of these most persistant and harmful hydrocarbons and (2)
the development of techniques to quantify rates for microbial
consumption and decomposition of aromatic and polycyclic aromatic
compounds in marine environments.
Specifically,
we intend to assay the weathering patterns and microbial activity in
anoxic, sulfidic sediments. The general approach is to incubate
hydrocarbon-contaminated seep sediments under controlled conditions
while assaying CO2/H2S/CH4 production,
sulfate consumption, and changes in petroleum composition.
We are using two distinct approaches to study the
intermediates and end products of hydrocarbon weathering. The
first approach is the use of radio-labeled substrates to assess major
products of microbial metabolism. The second technique involves
chromatographic technology designed to completely resolve the undefined
complex mixture typical of weathered petroleum, so-called
two-dimensional gas chromatography. We are collaborating with Chris
Reddy at Woods Hole Oceanographic Institution, who has helped develop
the GC×GC technology.
We have also performed our first experiments designed to
assess the rates of hydrocarbon weathering. One experiment
involved collecting a time series of surface slick samples at Shane
Seep, using the slick sampler developed by other MMS-funded scientists
(Liefer et al). Samples were collected at the seep and
‘down-slick’ and represent a rough time series.
A second experiment collected 5 L of
sediment from anoxic regions of Shane�s seep to be used in a
long-term incubation experiment. The sediment was
transported back to the lab, while being kept under environmental
conditions.
Analytical procedures:
Gases (CO2/CH4/H2S) are
quantified in the headspace using an agilent Micro GC/TCD at UCSB shown
in Figure 1. TCO2 in the aqueous phase will also be quantified
using the agilent Micro GC/TCD. Aqueous sulfur (~ sulfate) is
quantified by spectrophotometry and ICP-OES from the liquid phase
overlying the sediment at UCSB. Organic Acids are to be measured
by HPLC at UCSB. DIC will be measured at UCSB. Hydrocarbon
composition will be monitored using GC×GC at WHOI.
Figure 1.
Left panel shows an image of the agilent Micro GC/TCD.
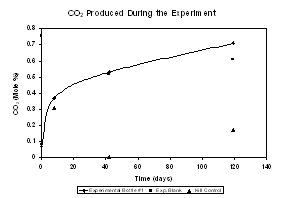
Figure 2. Preliminary results
are shown in the right panel for CO2 produced in the
experimental, blank and kill control bottles over the first 120 days of
the experiment.
Selected Results:
During the first 120 days of incubation kill controls
(autoclaved petroleum, sediment and seawater), experimental blanks
(sediment and seawater w/out petroleum) and experimental bottles
(petroleum, sediment and seawater) were analyzed periodically for
various markers/indicators of microbial growth. These experiments
have focused on quantifying CO2 in the head-space of the
incubation bottles, determining the δ13C of the CO2
in the head-space and an assay for the production of sulfide. The
isotope ratio mass spectrometer used for this experiment, along with
our initial results, are shown in Figure 2. Quantifying CO2,
the end-product of petroleum consumption by microbes, and analyzing the
δ13C of that CO2 will aid in determining the
extent of microbial activity. The production of sulfide should
indicate both that conditions are sufficient for microbial growth and
the presence of sulfate reducers which has been postulated to be a
necessary condition for microbial hydrocarbon oxidation.
Figure 3. Shows an image of the IR-MS used to determine
isotopic values for CO2 produced during the
experiment.

Figure 4. Right panel shows initial results for
the experimental (E#1), blanks and kill controls during the first 120
days of the experiment.
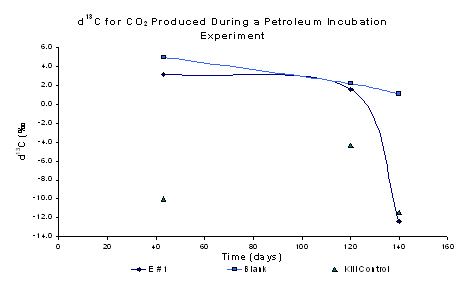
Analyses performed on the first 4 time series samples
show a steady increase in the quantity of the CO2 in the
experimental bottles. Results from experiments using the isotope
ratio mass spectrometer (IR-MS) on the CO2 in the head-space
show a decrease in the δ13C. Analyses performed on
December 18, 2003 show a δ13C value of approximately +3‰
whereas values from tests performed on experimental bottles on March
24, 2004 showed a decrease in the δ13C to approximately
-12.5‰. The δ13C value determined in the kill controls
has been static at approximately - 10‰. Sulfide assays show an
increase in sulfide produced in the experimental bottles from T=0 to
T=4. Aqueous sulfide concentrations have increased from below
detection to slightly less than 300 µM in the first 120 days of
incubation. Sulfide concentrations in the kill controls have
remained below detection throughout the experiment.
The surface slick samples collected for the ‘down slick’
time series did not contain enough oil to quantify and will be repeated
in the coming year.
Future plans:
We are in the process of analyzing samples from the
long-term incubation experiment, designed to broadly assay hydrocarbon
weathering patterns, including aromatics. The planned duration of
the experiment is 12-18 months. Samples will be sacrificed on a
monthly basis and assayed using the methods described above, including
the new GC×GC techniques developed at Woods Hole Oceanographic
Institution. We have acquired a Spectronic 20 spectrophotometer
for use in quantifying aqueous sulfate and to calculate sulfate
reduction rates in the experimental bottles. Since microbial
activity has been indicated, the first 5 time series will be shipped to
WHOI for GC×GC analyses.
|
[homepage]
|
[overview]
|
[researchers]
| [students
& staff]
|
[research] | [download reports]
|
|

